Competing COVID Vaccines?
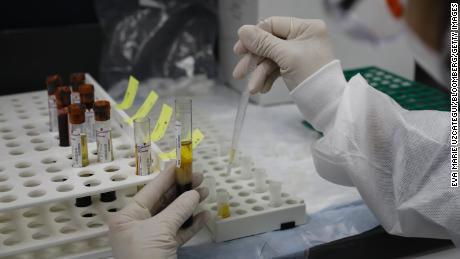
A health worker wearing a protective mask works in a lab during clinical trials for a Covid-19 vaccine at Research Centers of America in Hollywood, Florida, U.S., on Wednesday, Sept. 9, 2020. Drugmakers racing to produce Covid-19 vaccines pledged to avoid shortcuts on science as they face pressure to rush a shot to market. Photographer: Eva Marie Uzcategui/Bloomberg via Getty Images
What You Need to Know About the New COVID-19 Vaccines

On November 09, 2020, a company known as Pfizer, and their partner BioNTech, released a vaccine for COVID-19. According to them, this vaccine is more than 90% effective. But is there another company on the rise with a competing vaccine?
Just on Monday, November 16, a company by the name Moderna released a vaccine that is 94.5% based on their most recent trial. With these results similar too Pfizer’s, is there a race underway and how long before COVID-19 protocols are ceased?
To address the topic of competition, there has not been much reported. Overall, most sources are addressing the optimism in the air for the consistent progress being achieved. With the results of the COVID-19 vaccines being so close, we can only hope they both continue to succeed with their trials.
With the results of the trials from both vaccines so similar, it is important that we compare them and break down their composites. Both are classified as RNA vaccines, which means “part of the coronavirus’s genetic code is injected into the body” (New York Times). Injecting part of the genetic code allows the human immune system to train itself to combat the virus. Both vaccines also have to be stored in extremely cold temperatures with Pfizer’s stored at -75 degrees Celsius and Moderna’s at -20 degrees Celsius for up to 6 months (BBC). However, there are some different characteristics. The dose for the vaccine from Pfizer is 2 injections 21 days apart, while the Moderna vaccine is 2 injections 28 days apart. But before we decide which vaccine would work best for us there are important facts to remember.
We need to remember that both companies are still partaking in clinical trials for these vaccines. The results from both companies come from the 3rd stages, meaning the trials are not complete. The vaccines will also need FDA approval. We also need to remember that while the success of these vaccines is providing hope during the new wave of coronavirus, the pandemic is not over. We can not relax the protocol. It is still extremely important to social distance the best we can and to wear our masks.







